Today is a great day for science and humanity, says the American pharmaceutical company.
The first effective coronavirus vaccine could prevent more than 90% of people from getting Covid-19, a preliminary analysis shows.
Pharmaceutical companies Pfizer of the US and BioNTech of Germany said their experimental vaccine is more than 90 percent effective in preventing the COVID-19 disease, based on initial data from a large study, a major victory in the fight against the pandemic.
The high percentage of those who are protected makes the findings compelling.
Regulators have said they would approve a vaccine that is only 50 percent effective – protecting half of those who get it.
Their vaccine has been tested on 43,500 people in six countries and no safety concerns have been raised.
The companies plan to apply for emergency approval for the vaccine by the end of the month.
The vaccine – in addition to the treatment – is considered the best way to get out of the restrictions imposed on us already in the last few months of our lives.
There are about a dozen vaccines in the final stages of testing – known as phase 3 trials – but this is the first to show any results.
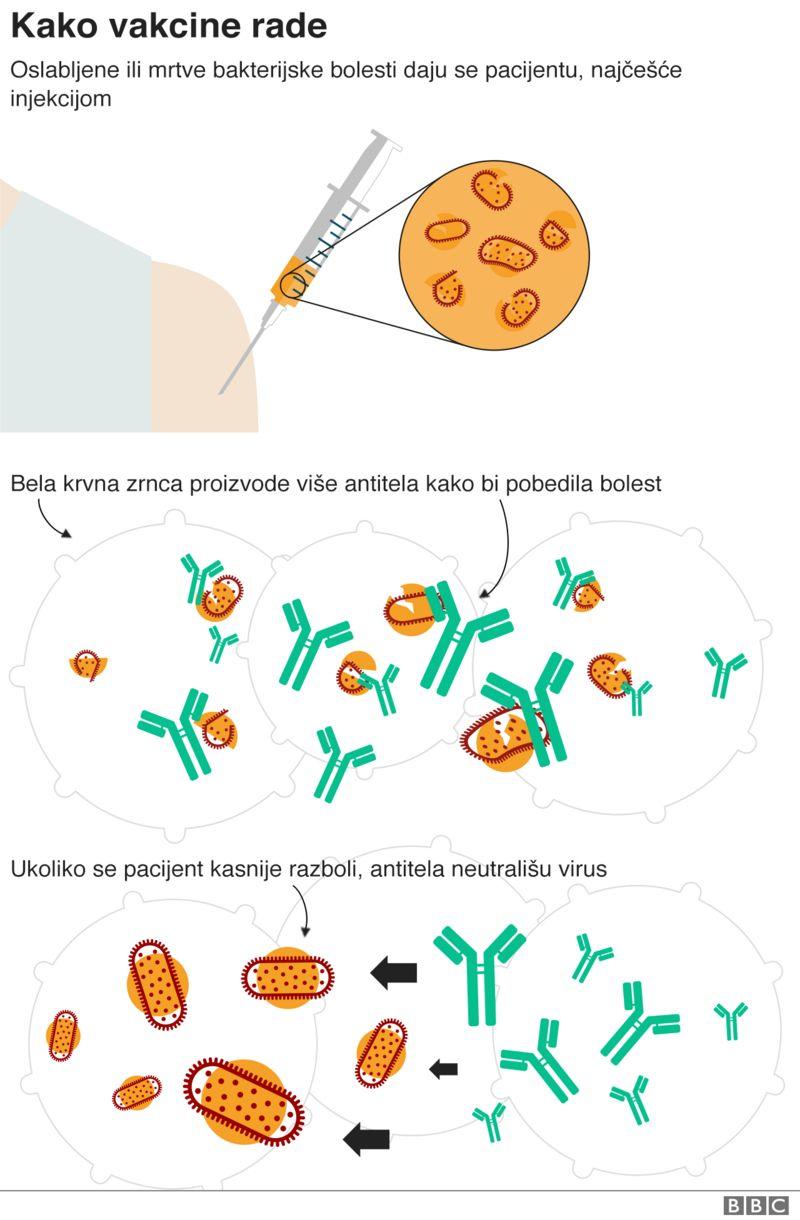
It uses a completely experimental approach – which involves injecting part of the virus’s genetic code – to train the immune system.
Previous trials have shown that the vaccine trains the body to make both antibodies — and another part of the immune system called T-cells — to fight the coronavirus.
Two doses are required, three weeks apart. Studies in the USA, Germany, Brazil, Argentina, South Africa and Turkey – show that it has been achieved 90% protection seven days after the second dose.
Pfizer believes it will be able to deliver 50 million doses by the end of this year, and around 1.3 billion by the end of 2021.
There are also questions about how long immunity lasts, and the companies have not presented a breakdown of the vaccine’s effectiveness in different age groups.
Dr. Albert Bourla, Chairman of Pfizer, said: “We are a significant step closer to providing people around the world with much-needed progress that will help end this global health crisis.”
Prof Ugur Sahin, one of the founders of BioNTech, described the results as a “milestone”.
The data shown is not a final analysis. It is based on the first 94 volunteers who tested positive for Covid – the precise effectiveness of the vaccine may change when the full results are analyzed.
The companies say they will have enough data to take their vaccine to regulators by the third week of November.
Until then, countries cannot begin their vaccination campaigns.
An official spokesman for the UK Prime Minister said the results were “promising” and that “the NHS is ready to start a vaccination program for those most at risk as soon as a Covid-19 vaccine is available”.
Taken from BBC News